Van der Waals Interactions: The Most Important Contact in CHL4 N-Terminal and Its adjacent Chains.
Study each interaction in details👇:
CHL4 N-terminal with NKP1, AME1, MCM21, OKP1, CSE4
Before exploring the basic properties of the protein, it is also important to analyze the interactions between chains[1].
Counting the number of hydrogen bonds, salt bridges, van der Waals forces between chains and the hydrophobicity of the interface are important to explore the affinity between them and to speculate that their function in the entire protein.
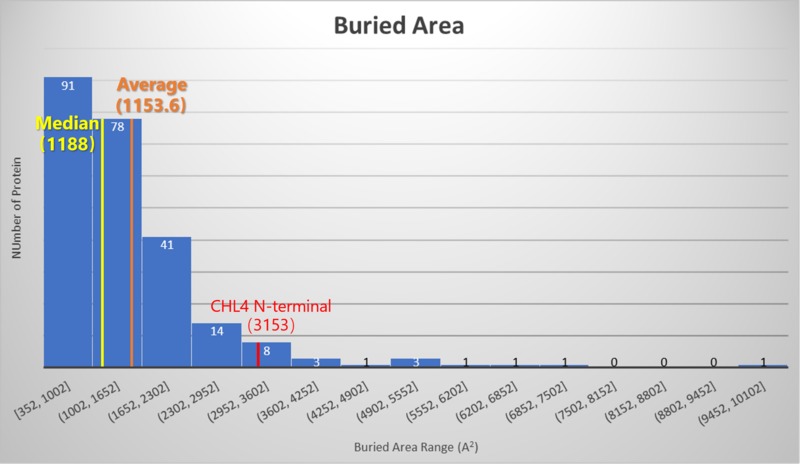
Hydrogen bonds, salt bridges and Van der Waals interactions all contribute to the protein-protein interactions of CHL4 N-terminal. However, only 1 salt bridge and 7 hydrogen bonds are involved in its interactions with other subunits of CCAN. Compared with its far above-average buried area, it is reasonable to assume that the Van der Waals interactions contribute most in the protein-protein interactions of CHL4 N-terminal.
*These two histograms are generated using the data from Ranganathan S. et al.
[1] Pierce M M, Raman C S, Nall B T. Isothermal titration calorimetry of protein–protein interactions[J]. Methods, 1999, 19(2): 213-221.
- - - - - - - -✂ - - - - - - - - - - - - - - - - - - - -- - - - - - - -✂ - - - - - - - - - - - - - - - - - - - -- - - - - - - -✂ - - - - - - - - - - - - - - - - - - - -- - - - - - - -✂ - - - - - - - - - - - - - - - - - - - -- - - - - - - -✂ - - - - - - - -
Go to the: ♦Function ♦Interfaces ♦Conservation of CHL4 N-terminal.
Go back to 🏠Welcome Page.