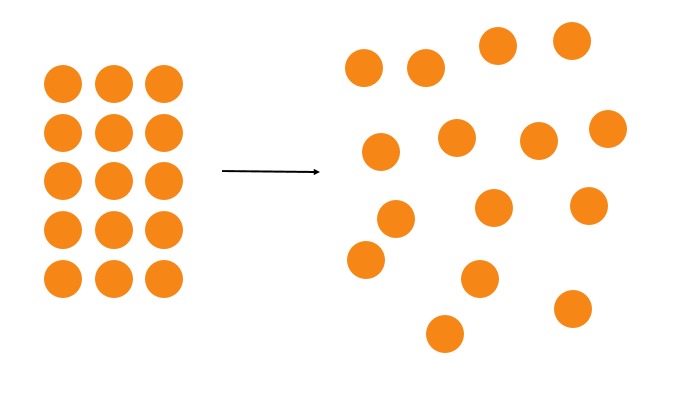
Entropy is often defined as a measure of disorder in a system, but what it actually measures is how heat is distributed. When two regions have different amounts of heat energy, heat spontaneously moves from the region with higher energy to the region with lower energy to evenly distribute the heat. Consequently, the degree of "order" of the initial system decreases and entropy (the degree of "disorder") increases.
The second law of thermodynamics states that the system tends toward disorder, which means that increasing entropy is always favoured.
The total entropy change of a system associated with binding is the sum of all entropic terms:
ΔS = ΔSsolv + ΔSconf + ΔSr/t
ΔSsolv - entropy change upon release of solvent molecules due to binding
ΔSconf - entropy change due to change in conformational freedom upon binding
ΔSr/t - entropy change due to change in rotational and translational degrees of freedom upon binding.1
For the rest of this virtual exhibit, we will focus on ΔSsolv and ΔSconf. Further explanations of these two terms are on the next page.
Reference
1. Du, X.; Li, Y.; Xia, Y. L.; Ai, S. M.; Liang, J.; Sang, P.; Ji, X. L.; Liu, S. Q., Insights into Protein-Ligand Interactions: Mechanisms, Models, and Methods. International Journal of Molecular Sciences 2016, 17 (2), 34.