Relationship between Entropy and Binding Affinity:
ΔG = ΔH - TΔS
Ka = e-ΔG/RT
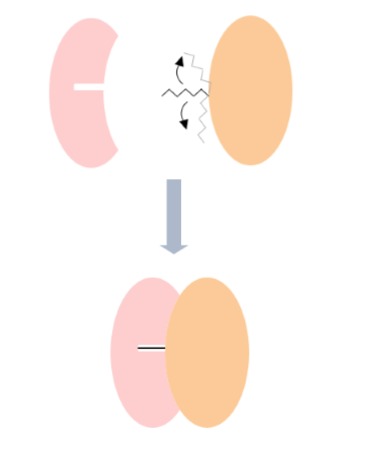
Hydrophobic Interaction
As we all know, hydrophobic molecules do not interact with water.
When proteins with a hydrophobic exterior are suspended in aqueous solutions, water molecules surround the hydrophobic exterior to form a shell. To form this shell, water molecules are fixed in a certain orientation around the hydrophobic surface to maximise hydrogen bonding.
When two hydrophobic surfaces come together, water molecules surrounding the hydrophobic residues are released. Because these water molecules are no longer fixed in place, the entropy of the system increases.
Hydrophobic interaction is considered as the most important interaction stabilising the folded conformations of proteins.1
As ΔS increases, ΔG decreases and Ka increases.
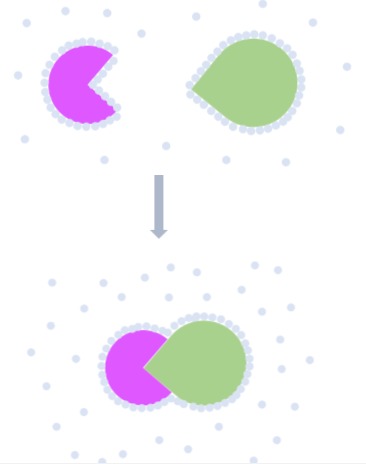
Conformational Entropy
A monomer often has more conformational freedom than a complex. For example:
1. The side chain of residues on the surface of a monomer has free range of motions.
2. Certain structural elements may be flexible (e.g. loops).
When binding occurs, these flexible structures are arranged in a certain conformation to maximise bonding interactions. This leads to loss of conformational freedom, resulting in decrease of entropy.2
As ΔS decreases, ΔG increases and Ka decreases.
References
1. Meyer, E. E.; Rosenberg, K. J.; Israelachvili, J., Recent progress in understanding hydrophobic interactions. Proceedings of the National Academy of Sciences of the United States of America 2006, 103 (43), 15739-15746.
2. Chang, C. E. A.; Chen, W.; Gilson, M. K., Ligand configurational entropy and protein binding. Proceedings of the National Academy of Sciences of the United States of America 2007, 104 (5), 1534-1539.