The Role of Stable Subcomplexes
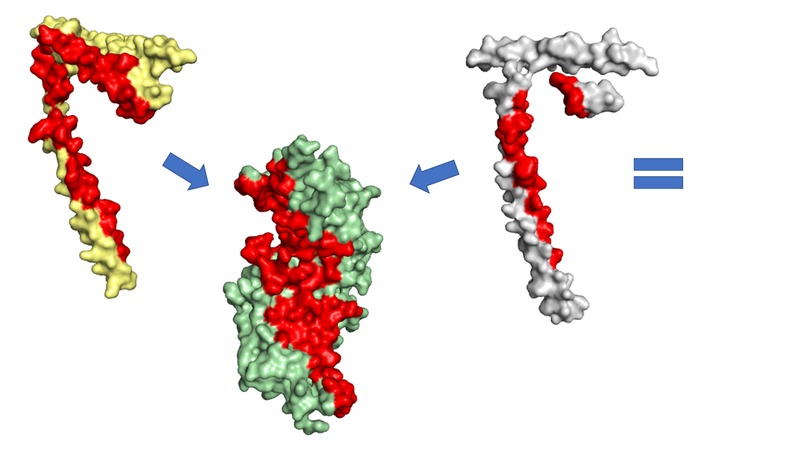
An example of the entropic effect upon the binding affinities between proteins can be observed in the yeast CCAN complex. Subunits such as MCM16, CTF3 and MCM22 have significantly higher binding affinities than other CCAN proteins; these subunits form the CENP-HIK subcomplex. In human CCAN, CENP-HIK is essential to the attachment of CENP-T to the KMN. This role may be reflected in yeast CCAN and would explain the relatively large binding affinities between CENP-HIK subunits provided in the following page. Effective complex formation due to low entropy loss between CENP-HIK subunits would ensure the attachment of CENP-T to the KMN, resulting in successful chromatid separation. Therefore the entropic aspects that determine the stability of the CENP-HIK complex may be essential to prevent aneuploidy.1 These include the large buried surface areas and hydrophobic interfaces that determine the solvation entropy. In addition, the loss in conformational entropy is limited as CENP-HIK is formed mostly of rigid α-helical contacts.
References
1. S. Kim and H. Yu, "Multiple assembly mechanisms anchor the KMN spindle checkpoint platform at human mitotic kinetochores", The Journal of Cell Biology, vol. 208, no. 2, pp. 181-196, 2015. Available: 10.1083/jcb.201407074.