How do Non-covalent bonds CONTRIBUTE to interface affinity?
For quick take home messages, spot 🔔🔔🔔
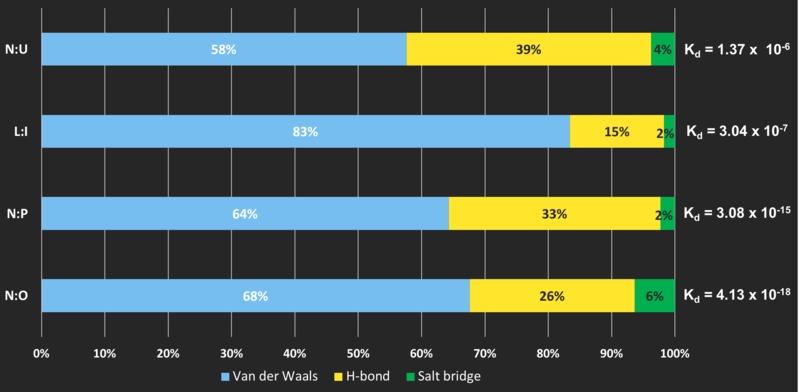
When ALL THREE TYPES of bonding are present...
Distribution of individual types of bonding is not crucial
No specific patterns are observed in the interfaces when all three types of bonding exists. There is a trend of increasing energetic contribution from Van der Waals’ forces and decreased energetic contribution from hydrogen bonds which resulted a higher affinity. This however, does not apply to interface IML3:CTF3 (L:I).
In general, the energetic contribution of salt bridges remain consistent.
To account for the differences in affinity, we suggested the following factors.
The actual number of non-covalent bonding involved matters
CHL4:AME1 (N:U) contains 4 hydrogen bonds, 1 salt bridge and 58 Van der Waals' interactions **
COMPARED TO
CHL4:CTF19 (N:P), which contains 15 hydrogen bonds, 3 salt bridges and 159 Van der Waals' interaction **
affinity INCREASES in 9-FOLD.
The interface shape may also affect their affinity
COMPARING interface CHL4:AME1 (N:U) and IML3:CTF3 (L:I): although CHL4:AME1 has a larger Buried Surface Area (BSA; 719.6 Å2 VS 546.4 Å2 for IML3:CTF3 and thus contains more bonding, having a FLAT interface makes it have a lower affinity (10X larger Kd) than that of IML3:CTF3, which interacts as CONVEX:CONCAVE.
Click on the shape description above to see them move ⇪
🔔🔔🔔
Looking solely at the overall distribution of all three types of bondings, no patterns were observed. However it must be noted that there are many more factors affecting the affinity of the interfaces.
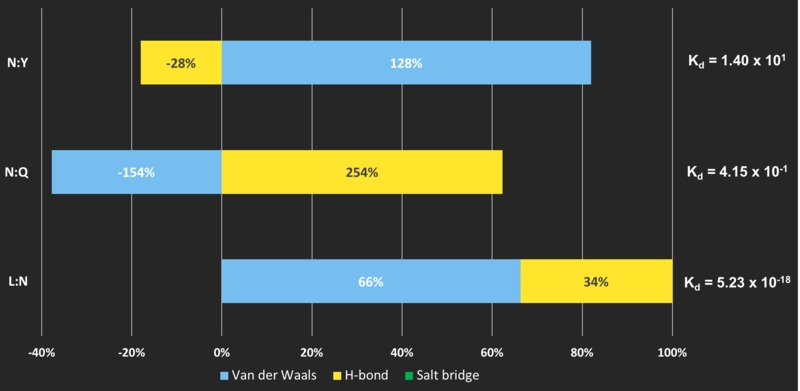
When Hydrogen Bonds and Van der Waals forces are present...
Negative energy contribution from non-covalent bonds could indicate unfavourable interaction
At interface CHL4:NKP1 (N:Y) and CHL4:OKP1(N:Q), hydrogen bonds and Van der Waals' forces respectively, negatively contribute to the energy at the interface.
Although another type of bonding compensates so that the subunit proteins are interacting, they have a remarkably low affinity compared to the rest of the interfaces Complex IML3:CHL4 (L:N) forms. This could indicate that both interfaces CHL4:NKP1 (N:Y) and CHL4:OKP1(N:Q) are not critical interactions in the CCAN conformation.
More evidence that CHL4:NKP1 and CHL4:OKP1 are not major interfaces
Indeed, this is further supported by the small number of bonding found and the BSA of these two interfaces.
CHL4:NKP1 (N:Y) - 1 hydrogen bond, 25 Van der Waals' interactions ** and BSA of 132.7 Å2
CHL4:OKP1(N:Q) - 2 hydrogen bonds, 10 Van der Waals' interactions ** and BSA of 211.8 Å2
🔔🔔🔔
There could be a specific type of bonding formed at the interface BUT NOT favouring protein-protein interaction, in this case, although contacts can still form, they are easily dissociated and less important than the others.
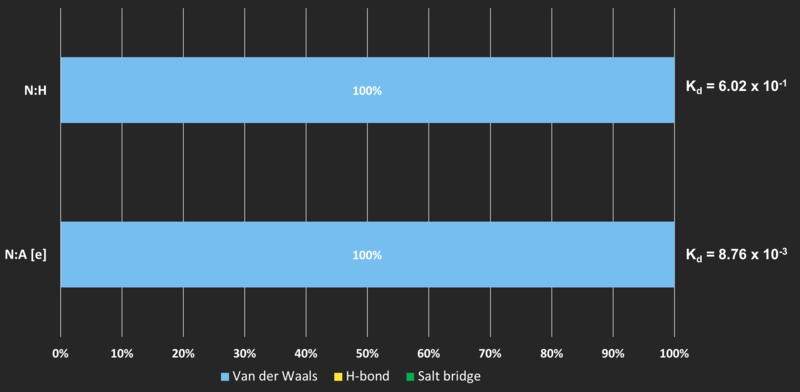
When ONLY Van der Waals forces are present...
ONLY when a lot of Van der Waals’ forces are present, the affinity is significantly affected
BOTH interface CHL4:MCM16 (N:H) and CHL4:CSE4 (N:A [e]) have extremely LOW affinities. Although increasing Van der Waals' interaction from 4 to 35 ** increases the affinity by 2-FOLD, there seems to be no effects in stabilising the interface.
Considering the biological roles of the CCAN complex and between these subunits, we could understand more about the presence of these interfaces.
Accounting for the low affinity
Tian et. al (2018) mentioned that not only is the assembly of CHL4 (Cenp-N) to CSE4 (Cenp-A) essential for chromosome segregation, but also needs to be dissociated at the G2 phase of cell division. Low affinity allows the separation to take place effectively.
On the other hand, while Cenp-L interacts quite heavily with CTF3 (Cenp-I) (Kd = 3.04 x 10-7), another subunit in the Cenp-HIK-TW complex, we presume that CHL4 (Cenp-N) touches MCM16 (Cenp-H) to allow the construction and stabilisation of the whole CCAN complex from various sub-complexes.
🔔🔔🔔
Only Van der Waals' forces at the interface may indicate a small contact. These can be unstable but essential for the CCAN complex to function properly in cell division.
** The number of Van der Waals' interactions are estimiated according to previous mention in OUR METHOD, results not shown.
Final Answer to the Hypothesis
The assumption that more types of bonds result in higher affinity seems to be UNTRUE. Have a look here for a bar chart summary.