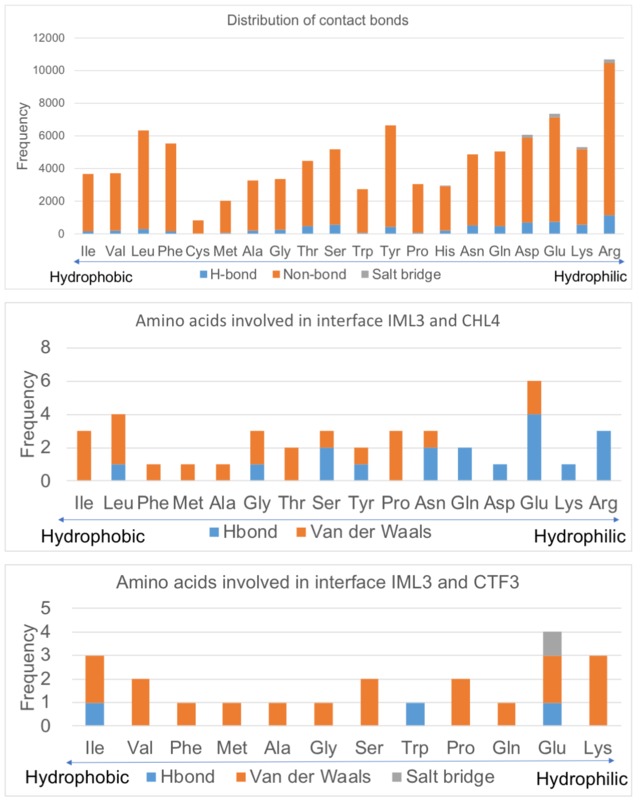
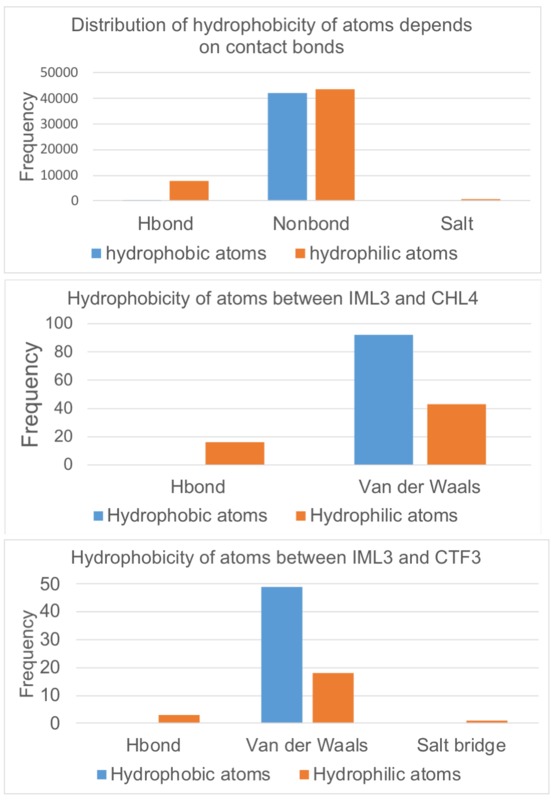
Hydrophobicity of atoms and residues
The figures below show that the H-bonds in the interfaces are composed mostly of hydrophilic residues and atoms, while Van der Waals' interactions are formed by hydrophobic residues and atoms. The hydrophobic effect may also participate in the formation of both IML3 complexes at their interfaces.